Lenacapavir: Di HIV prevention injection wey no dey safe for pregnant women, how e dey work?
Wia dis foto come from, AFP via Getty Images
Nigeria govment don begin rollout a new long-acting HIV prevention injection wey be Lenacapavir, but say pregnant women no fit use am.
Di Director-General of di National Agency for di Control of AIDS (NACA)Temitope Ilori na im tok dis one say di rollout go begin for eight states.
Di states na Anambra, Ebonyi, Gombe, Kwara, Akwa Ibom, Cross River, Benue and di Federal Capital Territory FCT Abuja, as part of efforts to expand access to innovative HIV prevention options.
Why di drug no dey safe for pregnant women?
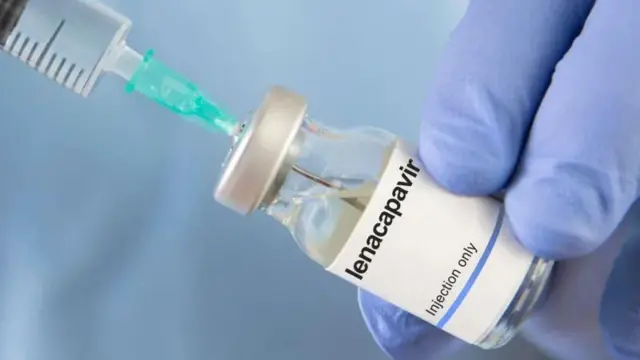
Di NACA DG Temitope Ilori explain say Lenacapavir na long-acting injectable antiretroviral drug wey Gilead Sciences develop for HIV prevention and na evri six months dem dey give am as a pre-exposure prophylaxis (PrEP) as dis na anoda alternative to daily oral pills, especially for pipo wey no fit adhere to di regular time to dey take am.
She note say while global trials suggest say di drug dey largely safe, enof evidence wey support to use am during pregnancy no dey and so pregnant women no fit use am sake of insufficient safety data.
"Even though LenPrEP dey considered to dey very safe based on global studies, e no dey recommended for pregnant women. E dey important to emphasise dis point becos dis na preventive intervention, not a treatment." She tok.
Di NACA DG describe Lenacapavir as ogbonge addition to di HIV prevention tools but give caution make pipo no overrate am as e no dey protect against oda infections.
"We must stress say e no dey prevent sexually transmitted infections, unwanted pregnancy or oda infections like hepatitis so pipo must kontinu to take comprehensive precautions."
She add say though di drug don gain global attention for di potential to simplify HIV prevention especially among high risk populations, e no be a standalone solution and must be used along wit oda preventive measures such as condom use and regular testing.

Wia dis foto come from, National Agency for the Control of AIDS (NACA)
Nigeria and di HIV burden
According to NACA, Nigeria rank fourth on HIV burdens globally wit an estimate of 1.9 million pipo wey dey live wit HIV.
While access to antiretroviral therapy don improve in recent years, gaps in prevention still dey persist, particularly among young pipo and key populations wey dey face such challenges as stigma, low awareness and inconsistent access to prevention services wey dey contribute to drive new infections.
Di rollout of dis drug wey dem go dey give twice yearly go make a significant step to end HIV/AIDS as a public health threat by 2030, through strengthened prevention, treatment and care services across di Kontri.
NACA emphasise say di drug no be cure but e only dey complement existing prevention options wey include oral pre-exposure prophylaxis and long acting injectable cabotegravir, wey go give choice to pipo wey dey at risk, as such, na prevention option for HIV-negative pipo wey get substantial risk of infection.
About 52,000 doses of Lenacapavir already don land Nigeria as di first batch dey ground a d preparations dey on for di national launch and facility-level rollout across di selected states.
Minister for State for Health and Social Welfare, Iziaq Salako say Nigeria dey selected as one of di nine adopter kontris by di Global Funds after dem express interest for March 2025 and receive approval.
Di phased approach for di rollout of dis drug na to ensure proper monitoring of safety outcomes bifor a nationwide expansion to oda states.
Di National Pre-Exposure Prophylaxis Implementation plan 2025 to 2028 go guide scale-up wey go cover service delivery, supply chain, financing, and community engagement.
E cut strong warning say becos di drug now dey available not be reason to encourage risky behaviour so make pipo no loose guard.